This article was originally published – The High Cost of Public Reporting – Medscape – Oct 03, 2017.
In an age where big data is king and doctors are urged to treat populations, the journey of one man still has much to tell us. This is a tale of a man named Joe.
I knew none of these things when I met Joe in the emergency department on a Sunday afternoon. I had been called because of an abnormal electrocardiogram—the ER team was worried he could be having a heart attack. Joe was gruff, short with his answers—but clearly, something just wasn’t right. He was only 54 but had more problems than the average 50-something.
Kidney failure many years prior meant dialysis sessions three times a week, and most recently, abnormal liver tests had revealed the presence of the early stages of cirrhosis from hepatitis C. Yet Joe continued to live an active life—with only a tight circle of family and friends aware of the illnesses beneath the surface.
But on this particular day, it was readily apparent Joe was not well. It didn’t take long to figure out that Joe had an infection somewhere. Review of his ECG and telemetry revealed a left bundle branch block and atrial fibrillation, but no evidence of a heart attack. A bedside ultrasound demonstrated a heavily calcified aortic valve, but little else. I told Joe and his anxious wife that he needed to be admitted to the hospital to find where the infection may be coming from.
I suspected that one of his dialysis sessions had been the conduit to allow bacteria into his bloodstream. I told the admitting team that if no other source of infection became immediately obvious, he would need an ultrasound to more closely examine his aortic valve. That night, I was called by the overnight resident in the hospital because Joe had a low heart rate. He felt fine and his blood pressure was OK. I stopped a heart rate-lowering medication, hoping this may be the cause. The next morning, a review of his ECG telemetry demonstrated an ominous finding—episodic heart block.
The specialized conduction tissue that courses in close proximity to the aortic valve was conducting in a stuttering fashion because bacteria were eating away at precious cardiac tissue surrounding the aortic valve. Of the many places the body may harbor infections, this was the worst-case scenario—he had a cardiac abscess.
There is only one treatment for this: surgery.
Joe had too high a chance of making the institution look bad.
The following day brought a decidedly different tone. There was a brief discussion about a conversation with the hepatology team having changed the surgeon’s mind. A prediction score for patients with liver disease suggested there was a 50% chance he would be dead in 6 months regardless of his current life-threatening cardiac abscess. That seemed to be an overestimate, and hepatitis C is now a treatable illness, but regardless, I pushed back and responded that the chance he would be dead in 2 weeks with his current condition was 100%.
After some more parrying back and forth, the real reason for the cold feet emerged. Outcomes of cardiac surgery are publicly reported for all institutions and surgeons nationally, and in the state of Pennsylvania. After discussing the case with the other surgeons at the institution, the decision at this hospital had been made: Joe had too high a chance of making the institution look bad.
How did we get here?
Public Reporting
The desire for public reporting arises from an attempt to broadcast value to a public eager for information. The Institute of Medicine’s seminal report in 1999[1] suggesting that 98,000 patients die owing to preventable medical errors every year added a sense of urgency and necessity to the push to grade providers.
Public reporting actually began more than 30 years ago—in 1984—when the Health Care Financing Administration (HCFA), now known as the Centers for Medicare & Medicaid Services (CMS), began to publicly report the hospital mortality rates of Medicare patients. The agency identified 269 hospitals that were outliers with regard to death rates. Although the analysis attempted to control for a variety of risk factors, it was heavily criticized,[2] and eventually the HCFA stopped publishing the data.
There was no turning back, though. New York state and Pennsylvania soon followed suit and started publicly reporting cardiac surgery outcomes. The group in charge of reporting in Pennsylvania was the Pennsylvania Health Care Cost Containment Council (PHC4), created by the Pennsylvania General Assembly with the charge of improving the quality of care and restraining healthcare costs.
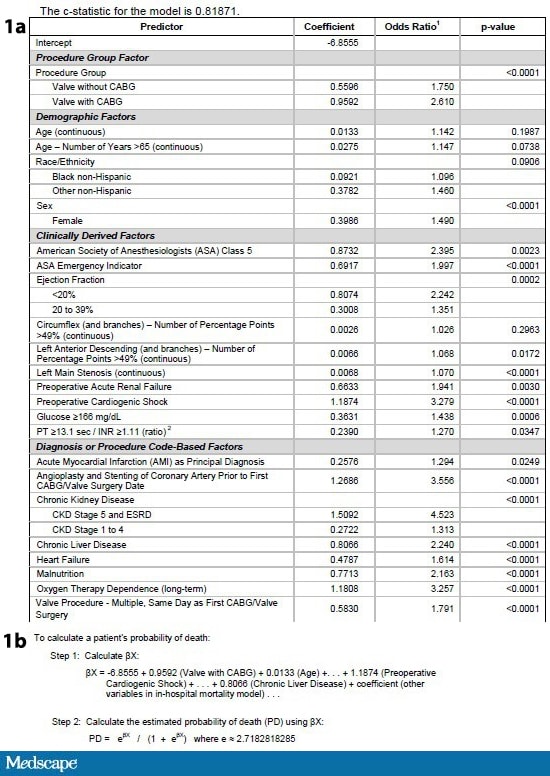
Figures 1a and 1b. Clinical predictor data table and calculation steps for: determining expected mortality. Source: Pennsylvania Health Care Cost Containment Council[3]
The final data are then presented for the public’s perusal in an easy-to-understand table with actual and expected mortality for every hospital and surgeon in Pennsylvania.
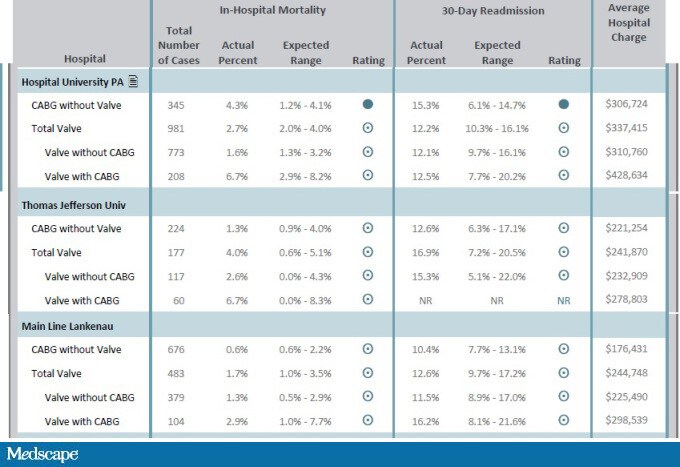
Figure 2. Observed and expected hospital mortality. Source: Pennsylvania Health Care Cost Containment Council[3]
Teaching to the Test
Proponents of public reporting will point to improved outcomes in the era of public reporting. In Pennsylvania, for instance, hospital mortality for coronary artery bypass graft (CABG) surgery dropped from 3.2% to 1.5%, and for valve surgery, it dropped from 5.2% to 2.7%.[3]
[T]he [cardiogenic shock] patients who survived a week were offered surgery.
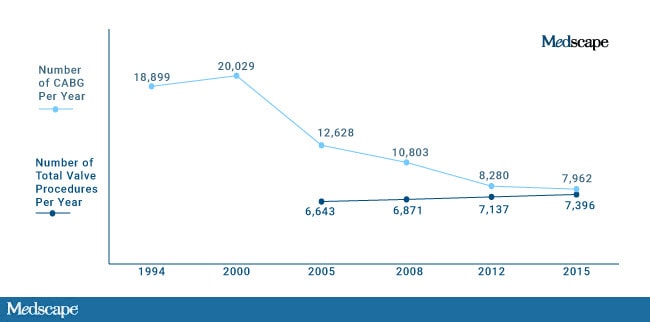
Figure 3. Volume of cardiac surgery. Source: Pennsylvania Health Care Cost Containment Council[3]
An optimistic conclusion to draw from the data is that there are fewer unnecessary procedures being performed and those that are being done are of higher quality in a safer environment. The more troubling explanation is that surgeons are operating less, and improved mortality relates to avoiding high-risk patients.
I spoke to a cardiac surgeon who had practiced in the public reporting era in New York state. He told me that “without a doubt” this was a consideration that affected whom he and his colleagues would take to the operating room. He spoke of how surgeon’s would “play the game” to improve their numbers by not necessarily denying the sickest patient, but rather waiting them out. He gave me an example.
One of the stronger clinical predictors that affected expected mortality was cardiogenic shock. A common approach by surgeons was to place these patients in the cardiac critical care unit (CCU) or transfer the patient; the patients who survived a week were offered surgery. The cold calculus makes sense—the expected mortality of the patient continued to be high, but clearly, waiting allowed selection of a lower-risk group of patients.
I was skeptical of finding published data to support what I had heard, because I frequently find that data sets simply aren’t granular enough to represent clinical practice. In this particular case, I was rescued by the SHOCK registrythat compared New York patients and non-New York patients who presented in cardiogenic shock.[4]
Table. Differences in Treatment Time of Patients With Cardiogenic Shock in the SHOCK Registry[5]
| New York Patients (n = 31) | Non-New York Patients (n = 49) | PValue | |
|---|---|---|---|
| Median time from shock to CABG (h) | 101.2 | 10.3 | <.001 |
| < 72 hours from shock onset (%) | 32.3 | 77.5 | <.001 |
| CABG = coronary artery bypass graft | |||
The results are stunning. As highlighted in the above table, the time to cardiac surgery in patients presenting with a heart attack and shock in New York state was different by almost 4 days!
The footprint of risk aversion is found even in the published data supportive of public reporting. Peterson and colleagues[6] found declining CABG mortality in New York state with no apparent decrease in access, but troublingly could not elucidate a mechanism for the decline in mortality. If the point of profiling hospitals and surgeons as good or bad was to direct more patients to high-performing centers, New York state officials found no evidence of migration of patients from high- to low-mortality hospitals.
Surveys of cardiac surgeons bear out that en masse, public reporting has markedly altered practice, primarily by denying patients surgery.[5]
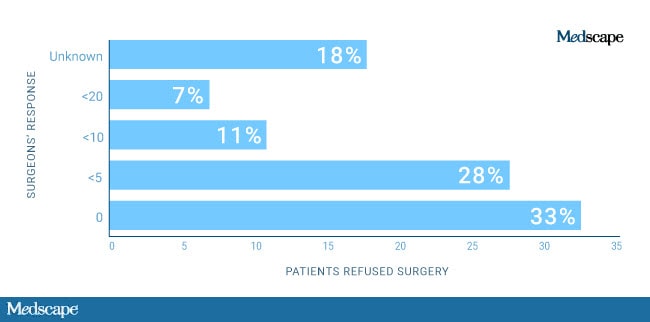
Figure 4. How many coronary artery bypass graft (CABG) patients were refused surgery with the Cardiac Surgery Reporting System being an integral part of the decision? Source: Burack JH, et al.[5]
It used to be that a decision on surgery was between surgeon and patient. Risks were outlined, and a decision arrived at. The patient and doctor were in it together. Surgeons operated on high-risk patients as long as they felt comfortable that the patient and family understood the risks. This construct exists no more. Especially in an era where surgeons are employed by healthcare systems, the surgeon is now beholden to masters who never step into hospital rooms. Surgeons with high mortality rates that make the institution look bad, face serious repercussions, and even worse, put future employability at risk.
Shared decision-making is a joke—not because decision-making isn’t shared, but because the shared decision is among surgeon, risk score, and hospital system. The sickest patients don’t have a choice anymore.
Back to Joe

Joseph Carrigan and his wife, Debra.
And so it was with Joe. Faced with certain death, he wanted to live, but I could not find a surgeon at my institution to operate on him. The hyperefficient healthcare system moved quickly in this case. The plan from the CCU team was now hospice/palliative care. I couldn’t stomach it. I told Joe that I was going to find a surgeon who would give him a shot. So I called a center that did the most valve surgeries in the city. This wasn’t the first time in my years in practice that a patient of mine had been turned down by a surgeon at a local institution. Smaller-volume centers are even more susceptible to risk aversion because their smaller volume amplifies the effects of bad outcomes. The largest center in the city had bailed out my patients before.
This time was different. Unbeknownst to me, the nationally renowned busiest center in the city had been taken to task in the Pennsylvania public report because they had been found to have worse than expected mortality. The expected mortality was 1.2%-4.1%, and the observed mortality was 4.3%. Of 345 patients who underwent bypass surgery, 15 died—one fewer death would have put the center in the expected range. To rub salt in the wound, the local paper ran a story on the report and quoted a competing health system’s surgeon as noting that the difference could relate to a minimally invasive surgery they did more of. Not mentioned was the fact that the smaller competing health system routinely sent their sickest, most complex patients to the larger academic center.
So, the answer again was no. Undeterred, I called Johns Hopkins next. The cardiac surgeon patiently listened to my story—and inquired why no hospital in the city would operate on him. I told him about public reporting, and he in turn told me about Maryland’s global payment system. Maryland hospitals were not paid per admission anymore; they were paid a per capita amount related to the number of patients attributed to them. The positives are that hospitals are incentivized to set up systems to keep patients out of hospitals. The downside is that they have a powerful disincentive to take on a complicated, high-risk, out-of-state patient like Joe. I was told bluntly that Maryland was not paying for this.
I discussed the case with a prominent cardiothoracic surgeon who said explicitly, “Politically impossible to do this case… Honestly, the big picture: He is a casualty of the public reporting system we have in the US and Pennsylvania, where risk aversion is always with us. It’s kind of sad. There are consequences to all the decisions society makes.”
Consequences. Trade-offs. This is decidedly not what the public hears. The public hears such things as the “triple aim”—improve patient experiences of care, improve the health of populations, and reduce the per capita cost of healthcare. You can have your cake, and eat it too! But there is a cost to all this, and it is a cost borne by our sickest, most vulnerable patients.
There are so few of these patients relative to the total that they won’t move the needle when it comes to the numbers that population health devotees care about, such as life expectancy and overall cardiac mortality. But the impact of this culture shift extends beyond the smattering of patients affected.
Risk Averse
America has long been the envy of the world when it came to innovative surgical techniques. The landscape for much of American history has consisted of brash physicians willing to push the envelope in dying patients with impossible odds. Bennie Solis was one such 3-year-old, dying of an irreversible liver disease when he was operated on by Thomas Starzl in 1963. Starzl thought he had perfected the technique of transplantation in dogs before attempting the feat—but he was wrong. Bennie bled to death on the operating table.
Starzl and the surgical team were devastated, but the lessons learned with Bennie’s death increased the chances of success for the patients that followed. This is true of the history of every new surgical procedure attempted. Innovation requires risk-taking, not risk aversion. It is difficult to imagine a man like Starzl taking on the sickest of the sick patients at Johns Hopkins today.
The sad part is that the age of penury driving this behavior is one where overall healthcare spending continues to accelerate, to the tune of $3 trillion annually. Our healthcare overlords may save some dollars on Joe, but won’t blink when spending billions of dollars on healthcare accountants, data entry clerks, hospital coding specialists, and any number of low-yield primary care preventions geared to the worried well.
Some think the solution is simply better risk-adjusting, or exclusions for centers performing at the frontiers. Perhaps. Or throw the whole system out and focus on the rotten apples among us. I don’t know. I do know that however well-intentioned the practice of public reporting may be, the consequences may be severe.
I couldn’t find a surgeon who would operate on Joe. So he died.
He never made it home to play his guitar.

Joseph Carrigan.
The pictures and the story are presented with the permission of Joe’s wife, Debra. Ever gracious, she hoped his story would teach us something.
