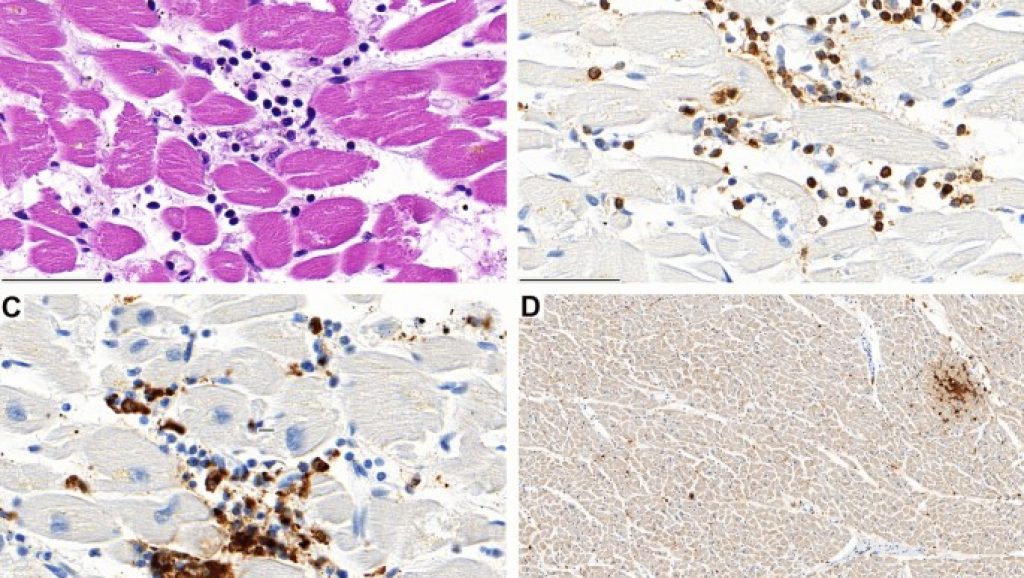
Between December 2020 and March 2021, the European Medicines Agency approved several vaccines on the basis of randomized, blinded, controlled trials: two messenger RNA-based vaccines—Comirnaty, BNT162b2 (Pfizer–BioNTech) and Spikevax, mRNA-1273 (Moderna)—both encoding the spike protein of SARS-CoV-2 encapsulated in lipid nanoparticles as the antigen and two vaccines based on recombinant adenoviruses
Are you a member?
Login
- About
- Advocacy
- Let’s Talk Medicine
- Resources
Resources
Let us be your go-to for all medical issues. Take a look at our helpful articles, webinars, research reports, papers and more.
- Health Policy, Campaigns and SubmissionsAdvocacy campaigns & submissions defending and promoting the doctor-patient relationship
- PodcastLet’s Talk Medicine
- Events and WebinarsConnect, learn, and engage with ADF members
- NewsroomHelping you stay across the latest news & announcements
- Articles Members OnlyExclusive insights, content, reports, papers and more
- Education Members OnlyExplore our library of material from Conferences and Presentations
- Child Protection
- Membership